Small Molecules to Treat Skeletal Disorders Caused by Integrated Stress Response
- Field
- Therapeutic Chemicals
- Reference No.
- IP00680
Background
- Skeletal disorders lead to physical disabilities and severely affect the quality of life in humans. Many skeletal disorders are caused by activation of integrated stress response arising from endoplasmic reticulum (ER) stress and chronic or prolonged mechanical stress.
- Current treatment options in these skeletal disorders are extremely limited and may involve controversial surgical procedures. For example, patients with congenital dwarfism, including Metaphyseal chondrodysplasia and other skeletal dysplasias, may be treated by growth hormone, but the treatment is not very effective. Intervertebral disc degeneration which causes low back pain (LBP), is very common in humans. Current treatment of LBP and disc degeneration is mainly surgical, often involving removal of the disc and spinal fusion.
Technology Overview
- This invention provides a novel approach to treat skeletal disorders caused by activation of integrated stress response using small molecules that inhibit the PERK pathway.
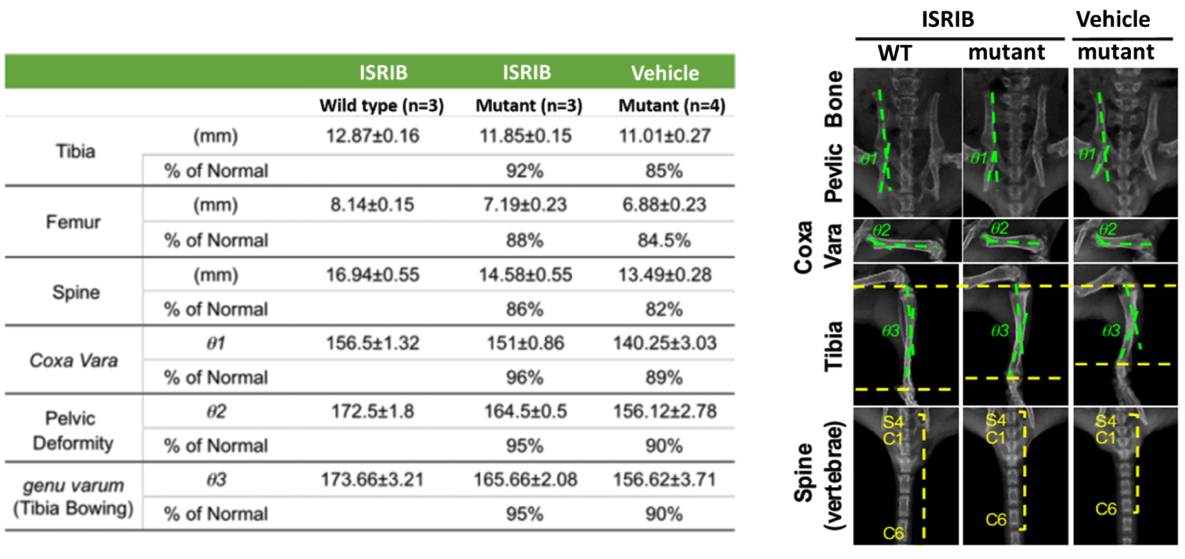
- A mouse model with phenotypes of congenital dwarfism and features of intervertebral disc degeneration were generated for preclinical study. The mutant mice have mutations in the gene encoding collagen X which induces integrated stress response.
- The mutant mice and wild type littermates were treated with ISRIB or vehicle daily. Radiographic analyses revealed that the dwarfism was ameliorated, and the skeletal deformities were greatly improved by the treatment of ISRIB. Treatment of ISRIB also eases the abnormalities of intervertebral disc degeneration syndrome in the mutant lumbar spine, too.
- Figure 1
Applications
- Therapeutic drugs to treat skeletal defects caused by activation of integrated stress response, for example, Metaphyseal chondrodysplasia and intervertebral disc degeneration.
- A mouse model with the phenotype of congenital dwarfism and intervertebral degeneration for developing potential interventions.
Patents
- PCT application No. PCT/IB2017/055783 was filed.