New Target for COVID-19 Diagnosis: the Nonstructural Protein 1 (nsp1) Gene
- Field
- Diagnostics
- Reference No.
- IP00947
Background
- SARS-CoV-2 can mutate quickly due to the intrinsic infidelity of viral RNA polymerase
- Currently, 3 major variants have been identified
- According to analysis by GISAID, mutations have been found in binding sites of primers and probes of published RT-PCR protocols
- The percent of genomes with mutation in the primer region was as high as 18%
- The global COVID-19 diagnostics market size is estimated at USD 84.4 billion in 2020 and is expected to expand at a compound annual growth rate (CAGR) of 3.1% from 2021 to 2027 (according to Grand View Research)
Technology Overview
This invention disclosed that nsp1 is a novel gene target for molecular detection of SARS-CoV-2. The addition of nsp1 for multiplex detection of SARS-CoV-2 can avoid false negative results due to mutations at the primers/probes binding sites of currently available RT-PCR assays.
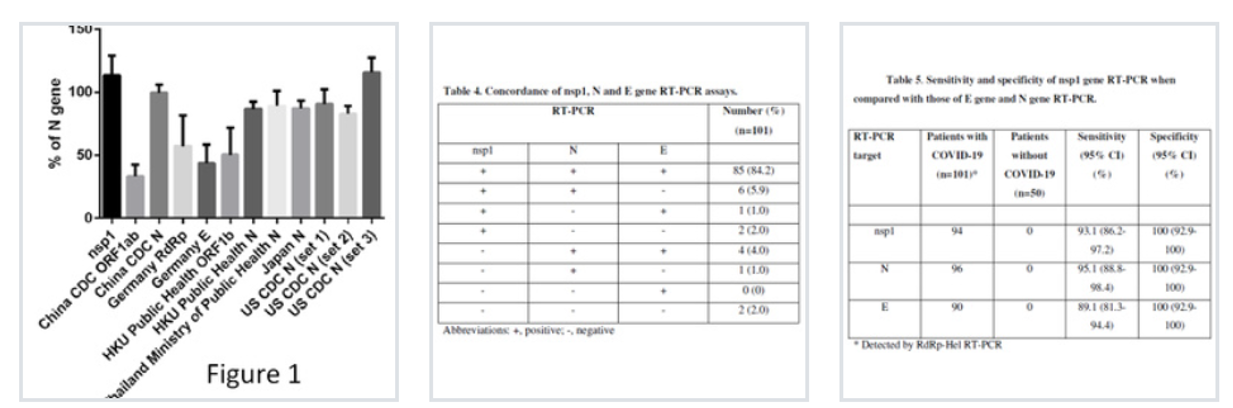
nsp1 real-time RT-PCR target region was expressed more abundantly than the gene targets of other published real time RT-PCR assay () and was more conserved in the 3’ end of the primer/probe region than that of N gene.
SARS-CoV-2 was detected by at least one of nsp1, N or E gene RT-PCR in 99 patients (98.0%), and 85 patients (84.2%) were detected by all 3 RT-PCR assays ().
The sensitivity was 93.1% for nsp1 gene RT-PCR, 95.1% for N gene RT-PCR, and 89.1% for E gene RT-PCR, while the specificity was 100% for all 3 RT-PCR assays ().
Benefits & Applications
Benefits
- Highly sensitive and specific
- Low limit of detection
- No cross reaction with other human coronaviruses or respiratory viruses
- Not affected by recombination events occurring between nsp1 gene and RdRp gene, since nsp1 gene is located in the 5’ end
Applications
- Primers/probes for COVID-19 diagnosis targeting specific sequences within NSP1 gene, for use in real-time RT-PCR
Patents
- US Provisional Application No. 63/013,282 filed 21 April 2020;
- PCT Patent Application No. PCT/CN2021/084357 filed on 31 Mar 2021
IP Status
- Patent application submitted