A Novel Broad-spectrum Antiviral for Treating COVID-19
- Field
- Therapeutic Biologics
- Reference No.
- IP00937
Background
Currently, the world is facing a pandemic situation due to coronavirus outbreak. As per WHO, the positive cases of COVID-19 across the globe rose to 416,686 with 18,589 deaths in the middle of March 2020. In addition, unavailability of a vaccine propels the COVID-19 diagnostics market growth. There is an urgent need for an effective antiviral against this virus.
Technology Overview
- This invention is about defensin like peptides (P9 and P9R) which exhibited potent antiviral activity against enveloped coronaviruses (SARS-CoV-2, SARS-CoV and MERS-CoV), the pandemic A(H1N1) pdm09 virus, avian influenza A(H7N9) virus, and the non-enveloped rhinovirus.
- P9R could significantly protect mice from lethal challenge by A(H1N1)pdm09 virus and showed low possibility to cause drug-resistant virus.
- Further mechanistic studies indicated that the antiviral activity of P9R depended on the direct binding to viruses and the inhibition of virus-host endosomal acidification, which provides a new concept that virus-binding alkaline peptides could broadly inhibit pH-dependent viruses. These results suggest that the dual-functional virus- and host-targeting P9R could be a promising candidate for combating pH-dependent respiratory viruses.
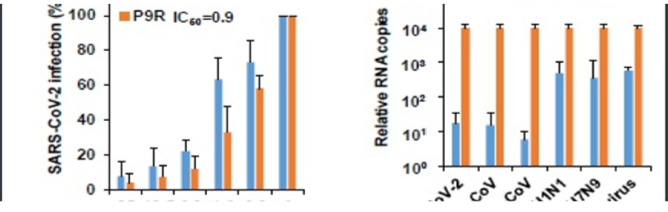
- Figure 1
(a) P9R could inhibit viral replication of COVID-19 (SARS-CoV-2); (b) The potent antiviral activities of P9R against viruses by measuring the viral RNA copies in supernatants at 24h post infection when viruses were treated by P9R or BSA
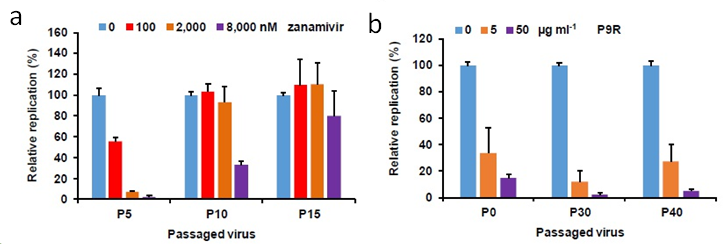
- Figure 2
Comparison study showed that P9R has low possibility to cause drug-resistance virus. (a) The antiviral efficiency of zanamivir against passaged A(H1N1) virus in the presence of zanamivir. (b) The antiviral efficiency of P9R against passaged A(H1N1) virus in the presence of P9R.
Applications
- Therapeutics for COVID-19 virus, other coronavirus and influenza viruses
Patents
- US Provisional Application No. 62/991,407
IP Status
- Patent application submitted